EZ 2.0 Point of Care Glycohemoglobin Analyzer
$350≥50Set/Sets
| Payment Type: | T/T |
| Incoterm: | FOB |
| Min. Order: | 50 Set/Sets |
| Transportation: | Ocean,Air,Land,Express,Others |
| Port: | Shanghai,Shenzhen |
$350≥50Set/Sets
| Payment Type: | T/T |
| Incoterm: | FOB |
| Min. Order: | 50 Set/Sets |
| Transportation: | Ocean,Air,Land,Express,Others |
| Port: | Shanghai,Shenzhen |
Model No.: A1C EZ 2.0
Brand: BioHermes
Types Of: Biochemical Analysis System
Place Of Origin: China
Warranty Service: 5 Years
After-sales Service: Return And Replacement, Free Spare Parts, Online Technical Support
Medical Device Classification: Class Ii
Glycated hemoglobin, also known as hemoglobin A1c (HbA1c), serves as an essential biochemical marker in the Diabetes Management, allowing for long-term glycemic control monitoring and the identification of potential complications. Studies such as the Diabetes Control and Complications Trial (DCCT) and the United Kingdom Prospective Diabetes Study (UKPDS) have demonstrated the direct correlation between glycemic control, as measured by HbA1c, and the risk of complications. It is for this reason that diabetes organizations worldwide recommend specific HbA1c targets based on DCCT/UKPDS HbA1c. To ensure consistency in clinical laboratory results, the National Glycohemoglobin Standardization Program (NGSP) was established to standardize GHB/HbA1c results according to the DCCT standard.
The BioHermes Smart Hba1C Device A1C EZ 2.0 is a handheld hemoglobin A1c device for diabetes management that utilizes boronate affinity chromatography, a patented technology in the USA, Canada, France, Germany, Great Britain, Italy, Japan, China, South Africa, Mexico, Brazil, Israel, India, and other countries. This clinic HbA1c device meets authoritative HbA1c standards, including those set by the International Federation of Clinical Chemistry (IFCC) and the National Glycohemoglobin Standardization Program (NGSP), and this Glycated Hemoglobin Device has been approved by CE and the U.S. FDA.
With room temperature transport and storage condition and simple operation steps, this innovative Hemoglobin A1c device make HbA1c Test more accessible and makes a contribution to the global warming by avoiding using cool room.

Features:
Accurate
NGSP and IFCC double certificates
Boronate Affinity Chromatography technology.no interference from HbF, HbE and other variable and unstable Hb
Accurate results with CV<3%
Convenient
Room temperature storage for all components
3 steps easy operation
Only about 3uL of capillary or venous blood sample
Fast
No preheat needed before testing
No manual calibration needed
Get the result within 5 minutes
Specifications:
Specifications
Technical Parameters
Testing Principle
Boronate Affinity Chromatography
Testing Item
Glycohemoglobin (HbA1c)
Testing Range
4%-14%
Precision
CV<3%(HbA1c:4.0%-6.5%)
Blood Sample
Fingertip blood or venous blood (EDTA Anticoagulation)
Blood Volume
About 3uL
Testing Time
About 5 minutes
Data Unit
Set in advance the data unit: NGSP%; IFCC mmol/mol
Voice Prompt
Voice prompt in whole process
Data Storage
1000 test results
Data Port
Mini USB data interface, can be connected withHIS/LIS system/thermal printer
Bluetooth Function
Optional
Power Required
AAA battery x 4
Analyzer Dimension
61.5mm x122.9mm x24.5mm
Screen Size
47mm x 32mm
Weight
112g (Does not include battery)
Operating Condition
Temperature: 10°C~40C; Humidity: 30%~70%
Storage Condition
Temperature: -10°C~50°C; Humidity: <80%
Simple Operation:
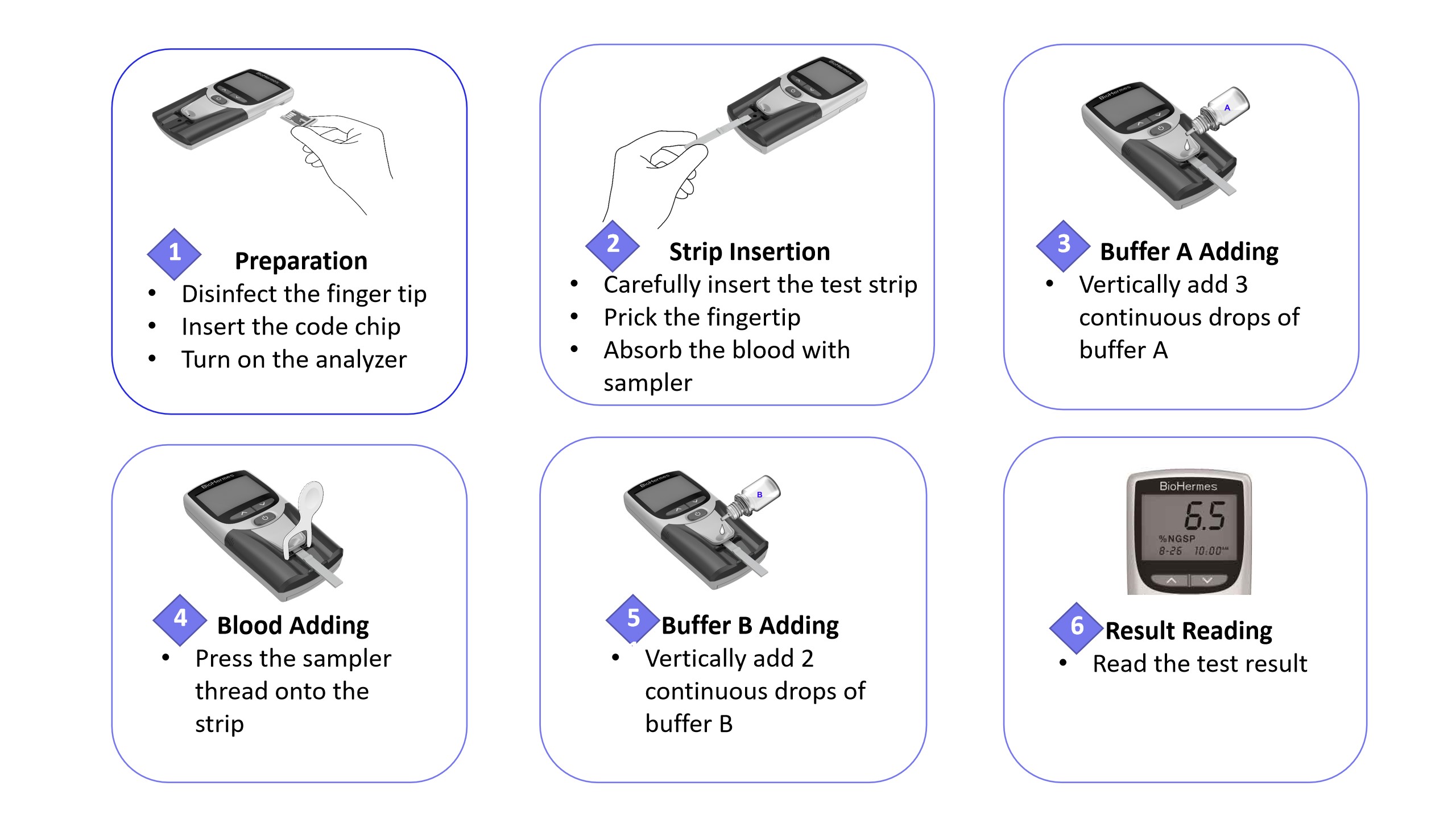


Privacy statement: Your privacy is very important to Us. Our company promises not to disclose your personal information to any external company with out your explicit permission.

Fill in more information so that we can get in touch with you faster
Privacy statement: Your privacy is very important to Us. Our company promises not to disclose your personal information to any external company with out your explicit permission.